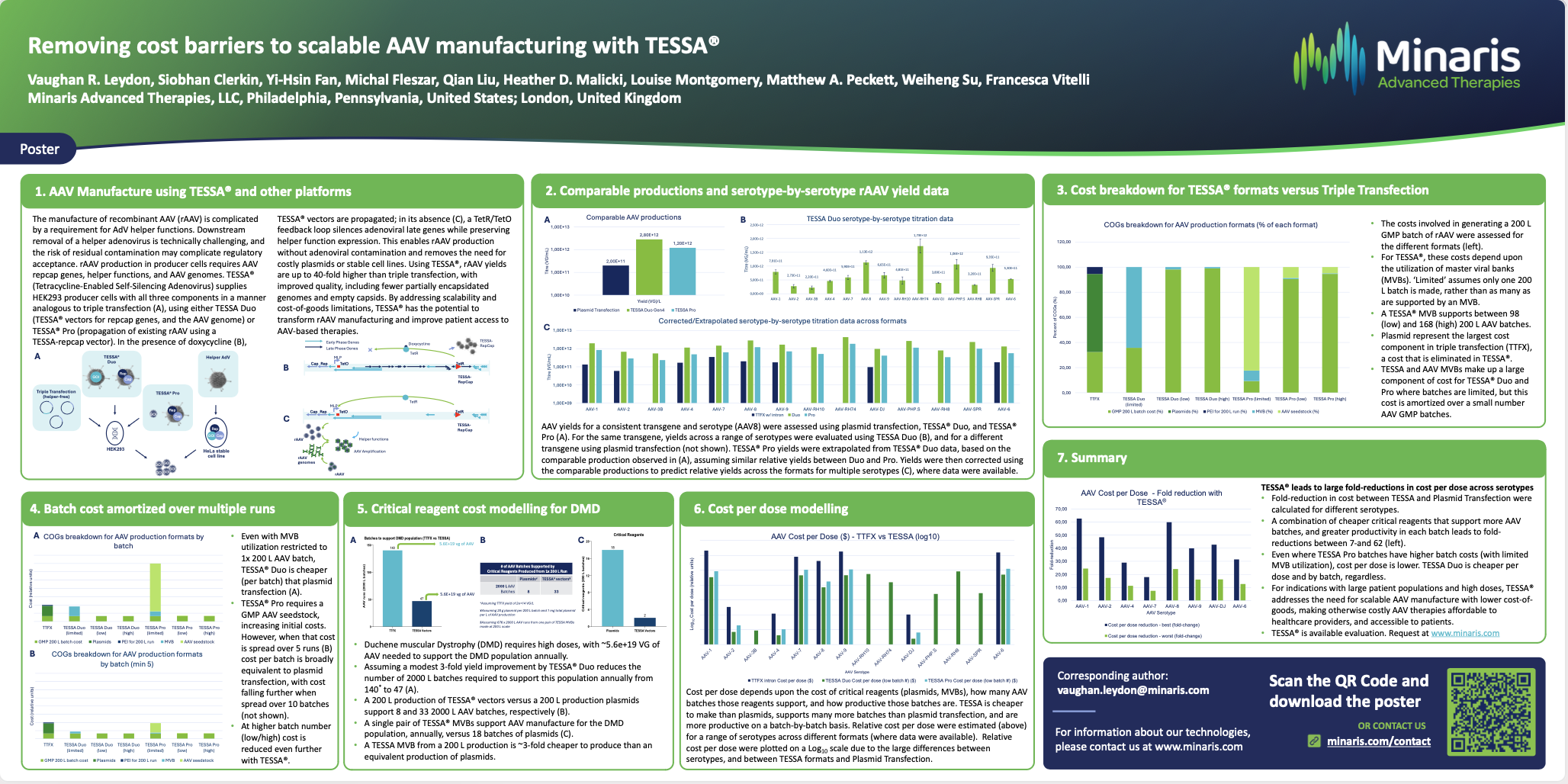
Adeno-associated virus (AAV) manufacturing continues to be costly due to high vector doses required for therapeutic applications and limitations of plasmid-based production.
Triple transfection-based production process, the industry standard, depends on large quantities of GMP-grade plasmids and reagents yet delivers low yields and titres, creating a major cost-of-goods barrier.
TESSA® (tetracycline-enabled self-silencing adenovirus) overcomes the cost-of-goods barrier in AAV manufacturing, delivering high yields, improved quality, and scalability, without adenovirus contamination.
This platform has the potential to drive down the cost-of-goods for AAV manufacture, transforming affordability and accessibility for gene therapy populations at scale.