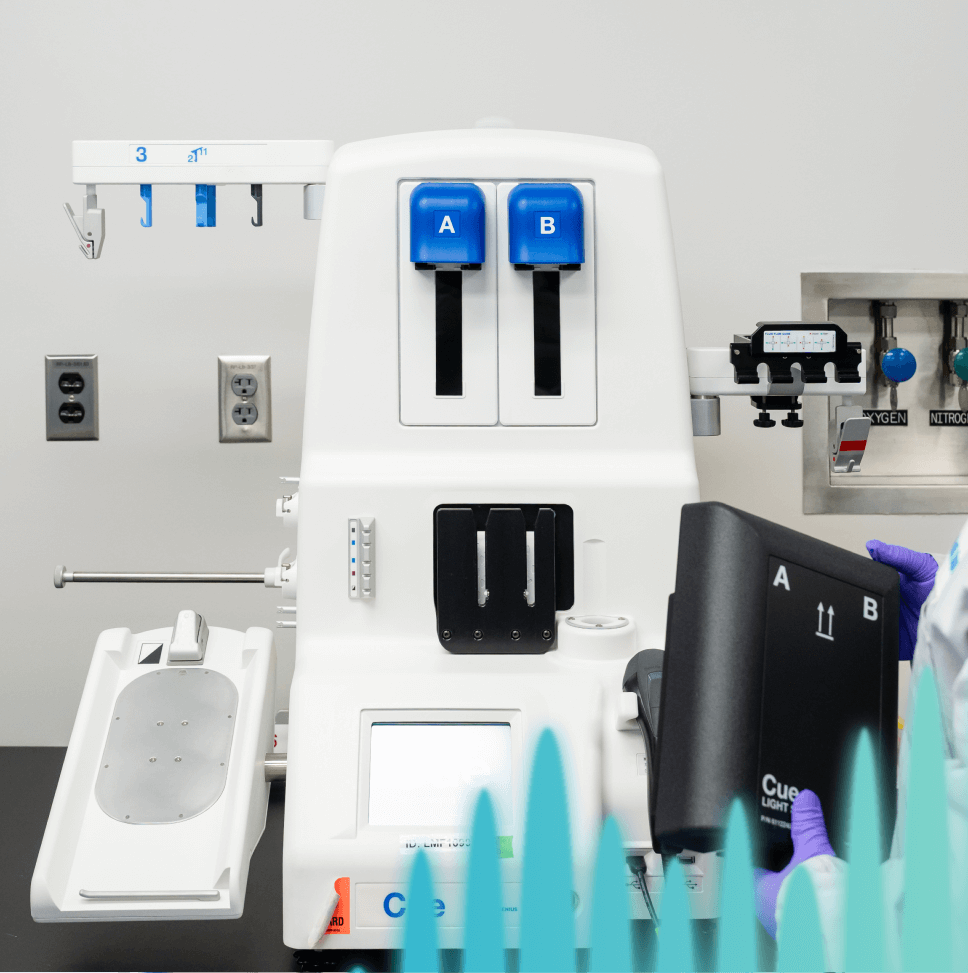
Cell therapy developers face persistent challenges, including long vein-to-vein times, high and variable costs of goods, deviations and non-conforming events, contamination, inconsistent yields, lengthy project timelines from contract signature to ready-to-use GMP drug product, and complex regulatory requirements.
Minaris Advanced Therapies has created technology platforms that directly address these barriers. With automated closed systems, optimized expansion protocols and scalable bioreactor solutions, we help innovators deliver consistent therapies that are cost-efficient and GMP-ready.